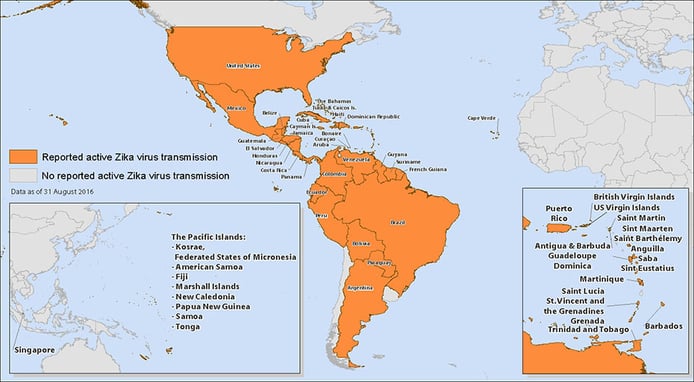
Zika virus continues to impact blood collection establishments and their efforts to keep the blood supply safe. Active transmission is occurring in the Americas, Oceania/Pacific Islands, Africa, and Asia. In the United States, the Food and Drug Administration (FDA) has released revised guidance regarding Zika virus testing, The FDA now recommends testing of all donated whole blood and blood components in the U.S. and its territories. Testing is to be performed with an investigational individual donor nucleic acid test (ID-NAT) under an investigational new drug application or with a licensed screening test, once one becomes available. Blood centers may also use FDA-approved pathogen reduction technology for plasma and certain platelet products.
Screening of all whole blood and blood components with investigational donor screening tests is already underway in Florida and Puerto Rico, based on confirmed reports of locally acquired mosquito-borne cases of Zika virus. Blood centers in other locations in the U.S. were given either 4 or 12 weeks to implement universal testing, depending on their proximity to areas with locally acquired cases of the virus.
According to the FDA, the revised recommendations will remain in place until there is sufficient evidence to determine that the risk of transfusion-transmission of Zika virus has changed. Additionally, since all blood will now be screened for the virus, the FDA is no longer recommending that blood establishments provide donor educational material or defer donors with risk factors for Zika virus. (These activities may be discontinued once the recommendations for testing or pathogen reduction are implemented.) Also, in order to help disseminate information and improve patient safety, AABB has established the Zika Virus Biovigilance Network, which tracks and maps U.S. locations where donated blood has tested positive for Zika virus.
Since 2005, Helmer Scientific has been contributing to advancing safety, availability, and usage of blood products locally and globally through the company’s generosity programs. We actively support the efforts of the International Blood Safety Forum initiated in 2012. Join us on March 24, 2017 for this collaborative initiative to improve access to safe blood around the world.
References
Image Credit: http://www.cdc.gov/zika/geo/active-countries.html
http://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/Blood/UCM518213.pdf
http://www.aabb.org/research/hemovigilance/Pages/zika.aspx



