
In December, at ASHP Midyear, Jeannell Mansur, Pharm.D., R.Ph., FASHP, FSMSO, Principle Consultant, Medication Management and Safety Joint Commission Resources, gave a presentation highlighting the 2018 Joint Commission Medication Management Updates.
She identified four medication management standards that were found to be non-compliant and described strategies to address each, described key features of the standard revisions pertaining to pain, and listed two new requirements in medication-related standards. The chart below examines the top noncompliant standards/NPSGs for hospitals. Storage and security of medications ranked high for non-compliance, yet again.
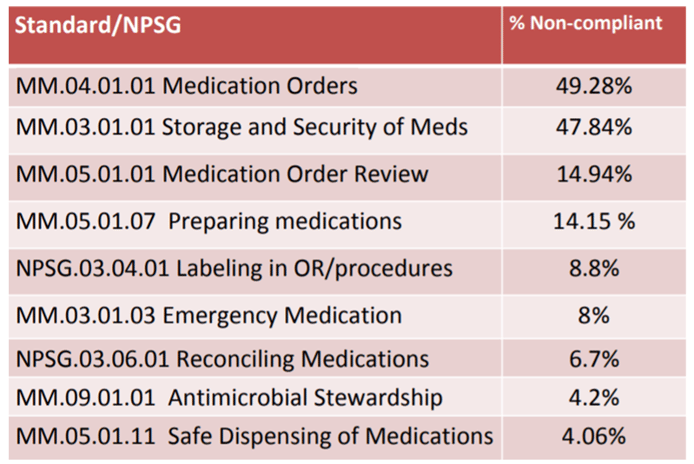
One of the major issues around medication storage and security pertained to controlled substances. All medications and biologics should be stored in a secure area to prevent diversion. These areas should be locked when necessary, in accordance to law and regulation. It was also noted that many hospitals are failing to address diversion.
Moreover, the hospital must prevent unauthorized individuals from obtaining medications. Particular attention needs to be paid to areas storing medications close to or at point of care, medications that are left unattended or in areas not considered secure, and in operating rooms not in use which store unlocked medications after hours.
More details related to storage and security, as well as the other items mentioned in the table above can be found in the slide deck provided by ASHP. The presentation also highlights the new requirments for pain assessment and management which became effective January 1, 2018. Follow the link below to review the full set of updates.




